
Heavy Metal Contaminations in Seafood
Laly, S.J. Heavy metal contamination in seafood. Smart Agripost – Fisheries Technology & Innovation.
The global population is depending upon seafood as a healthy diet choice because of its richness in high-value proteins, health-beneficial vitamins, minerals, and polyunsaturated fatty acids. Fish is also a primary protein source in most parts of the world. Even though fish supplies many health benefits, seafood can be compromised by different chemical contaminants which are harmful to consumers. Among the environmental chemical contaminants, heavy metals are reported as the most commonly occurring ones in seafood mainly lead, cadmium, arsenic, and mercury. These metal contaminants are coming to the environment from industrial sources and pollution. The toxic nature of heavy metals with high persistence in the environment along with bioaccumulation and biomagnification through the food chain makes a serious issue. Fishes can uptake and concentrate metals from water, other small fishes, and vegetation. Accumulation in the tissue can depend upon environmental concentration and exposure period. Also in an aquatic ecosystem fishes are the most important biomonitor for the estimation of heavy metal contamination (Rashed, 2001). It is significant that all the metals are not hazardous and toxic to fish and humans. While essential metals like copper (Cu), zinc (Zn), iron (Fe), iodine (I), and selenium (Se) play important role in biological
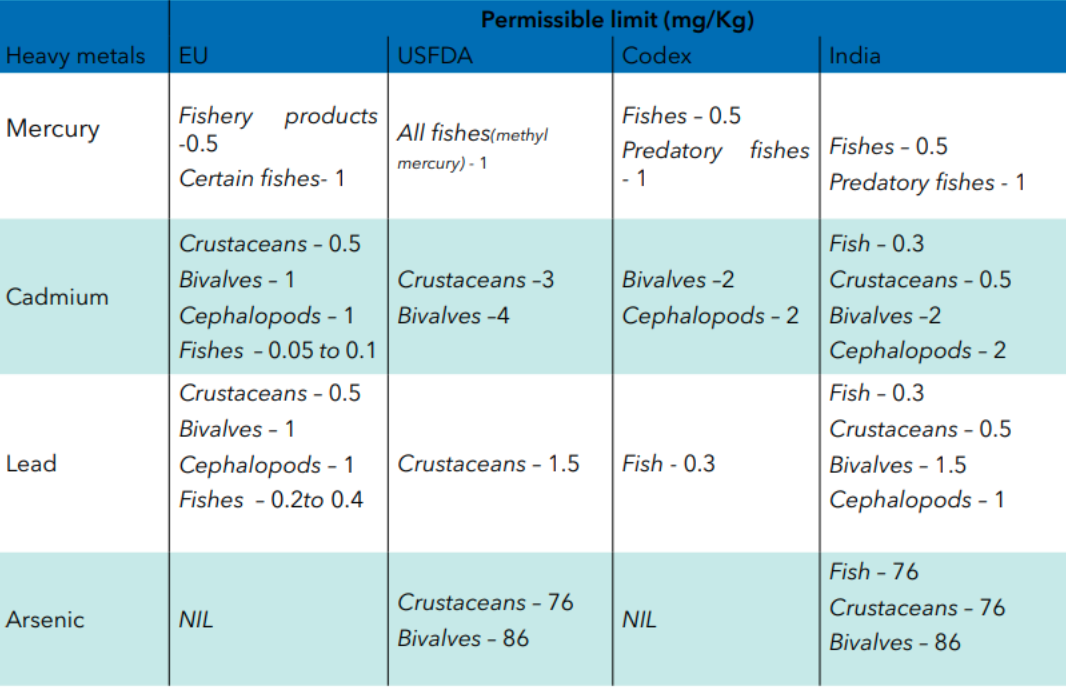
activity and can affect the human health condition during their deficiency or at higher concentrations. Cadmium (Cd), mercury (Hg), tin (Sn), arsenic (As) lead (Pb), etc generally make a harmful effect on living organisms. Based on the degree of toxicity, the maximum allowable limit (MAL) is determined which is a limit above which the consumers are exposed to harmful contamination. MALs of toxic elements have been decided by different countries and regulatory bodies based on the fish consumption pattern of specific population groups (Table 1).
Mercury
Mercury is the metal of most concern with respect to seafood consumption and human health as it is the most toxic among heavy metals. Fish is considered the most important source of mercury in the case of humans. Although forest fires and volcanic activity act as a natural source of Hg, the major part is from the anthropogenic origin such as from industries of paint, electrical equipment, battery, fungicide, wood pulping, etc. Even though the industrial use of Hg has decreased due to continuous awareness of Hg toxicity, the environmental Hg level is still on the higher side.
Speciation of mercury and toxicity
Elemental mercury (Hg) and mercuric ions (Hg2+) are the main forms present in the natural environment, but they will not accumulate in fish. It can be converted into other soluble forms such as inorganic and organic mercury. Mercuric chloride (HgCl2), mercurous chloride (Hg2Cl2), mercuric sulfide (HgS) are common forms of inorganic mercury that occur in nature. Although inorganic forms are non-toxic, they can be methylated to form organic mercury called methylmercury (MeHg) which is the chronic toxic form of mercury (Hempel et al., 1995). The process of methylation occurs either by photomethylation or microorganisms such as anaerobic sulfate-reducing bacteria, iron reducers, and methanogens. Among microorganisms, sulfate-reducing bacteria are the major contributors to Hg methylation in natural waters. Dimethymercury (DMHg) and ethylmercury (EtHg) are the other organic Hg forms other than methyl Hg. But MeHg is the main form absorbed into the food chain which is most toxic. Also, the half-life of MeHg is longer than inorganic Hg and has a good correlation with the percentage of MeHg on total Hg (Forsyth et al., 2004). The contribution of MeHg to total Hg is reported to be 10% in water, 15% in phytoplankton, 30% in zooplankton, and 95% in fish flesh (Watras and Bloom, 1992). Fish at higher trophic levels accumulate more amount of Hg than at lower levels due to its significant absorption. It creates a greater risk to consumer health as Hg can associate with protein fraction SMART AGRIPOST – FISHERIES TECHNOLOGY & INNOVATION of fish meat by binding with thiol group complexes. After consumption 95% of MeHg is getting absorbed through the intestinal tract. It can reach and accumulate in the brain via blood resulting in the loss of cells in specific brain areas and also affecting other target organs like the pituitary gland, liver, and kidney. The major symptoms of MeHg in humans are impaired vision and hearing, headache, paraesthesia, difficulty in movement, less coordination, fatigue, tremors, and ataxia (Grandjean et al., 2010). Prenatal exposure of children to Hg can result in developmental disorders. As seafood is the major source of beneficial polyunsaturated fatty acids consumption of predatory fishes like sharks, swordfish, king mackerel, etc can be avoided or consumed in smaller quantities. As per the international safety guideline, the maximum tolerable weekly intake of Hg is 1.6 µg/Kg body weight (EC 2006).
Arsenic
Arsenic (As) is widely distributed in the environment due to natural and anthropogenic sources. It has the ability to deposit in huge quantities in sediments of water bodies and reservoir systems. The major agricultural source of As is from industries manufacturing defoliants and pesticides. Arsenic is present in both organic and inorganic forms and the most toxic one among them is inorganic As which is stable and highly soluble and can be absorbed into the body easily. Organic form As cannot accumulate in the body and it is getting quickly excreted. Inorganic Arsenites (As3+) is more toxic and get easily absorbed into fish compared to arsenates (As4+). Although total As is measured instead of inorganic As (Arsinite), 90% of As present in fish and crustaceans is in nontoxic arsenobetaine form. While in the case of mollusks and algae it is present in the form of arsenocholine and arsenosugar respectively. The inorganic As trioxide is known as rat poison. It is reported that organic arsenic compounds and their metabolites can produce cytotoxic effects. The content of inorganic arsenic in seafood is very negligible generally lower than 0.2 mg/Kg in fish and shellfish. As per the European Commission Scientific Cooperation Project (SCOOP), the consumption of arsenic from marine origin foods may be in excess of 50%. As per EFSA, (2009a), seaweed has the highest total As concentrations in the marine food web. Shellfishes usually have a high content of arsenic than finfishes. Demersal fishes often contain higher As levels compared to pelagic fishes (Wu et al., 2014). As concentration in fish is normally less than 5 mg/Kg. The reference value proposed for intakes of inorganic As from seafood products is 0.3–8 mg/ kg BW/day (EFSA, 2009a).
The various symptoms of As exposure in humans include abdominal pain, vomiting, diarrhea, muscle weakness, and skin flushing. Continuous exposure to As can result in the deposition of As in keratin-containing tissues like nails, hair, and skin. In chronic cases, it leads to skin defects and cancer. International Agency Research on Cancer (IARC) has classified inorganic As a carcinogen to humans. Seaweeds can contain un-extractable or residual As linked with its structural components.
Cadmium
Cadmium (Cd) is a highly toxic heavy metal to all living organisms of plant and animal origin including microorganisms. Although Cd is a nonessential trace element in nature, it can bioaccumulate at hazardous levels in living organisms and it has no known beneficial function in humans. In addition to sources from industries manufacturing batteries, electroplating, plastic stabilizers, pigment, use of agricultural chemicals and fertilizers also contribute to the environmental contamination of water resources. It is also a naturally occurring nonessential trace element. It is a comparatively soluble toxic metal that can make many adverse effects including chromosomal damage. It can also affect the nervous system, kidneys, bones, lungs, and cardiovascular system. Cd can be long-term stored in the human being, with an average biological life of 17 to 30 years (Tokar et al., 2015). It is normally seen in the inorganic form in seafood with higher contents in shellfish especially in bivalves which may reach 10 mg/Kg. Bivalves can accumulate this toxic metal and act as suitable bioindicators for pollution monitoring. It can also bioaccumulate in plants, invertebrates, and vertebrates with a long half-life. Cadmium concentrates in freshwater and marine animals to concentrations hundreds to thousands of times higher than in the water ATSDR (2008). Biomagnification of Cd also happens in the food chain and increased concentration will reach the end of the food chain. The bioaccessibility of Cd from seafood products is high (>50%) (Gao and Wang, 2014). The tolerable weekly intake established by EFSA’s panel is 2.5 mg/kg BW (EFSA, 2009b) Lead Lead (Pb) is a highly toxic metal in the aquatic system in which fish are at the top of the food chain. It is also a naturally occurring metal, but anthropogenic sources from mining, manufacture of batteries, paint, pesticides, etc also contribute to environmental contamination. Inorganic lead occurs in soil, dust, and many consumer products such as lead carbonate, lead chromate, lead acetate, etc. The organic form of lead or tetraethyl lead is
extremely poisonous which can affect the brain and nervous system more severely than an inorganic form of lead. Lead poisoning or toxicity due to lead is common in people associated with its industrial usage. Although acute toxicity due to occupational exposure is comparatively less, chronic toxicity due to prolonged exposure at lower concentrations is common. The International Agency for Research on Cancer classified inorganic lead as probably carcinogenic to humans (Group 2A) in 2006. The major sources for dietary exposure include cereals, vegetables, and tap water. Pb accumulation in fish mainly occurs from contaminated water rather than diet, mainly inorganic form. In addition to fish, the other sources of Pb include bivalves, cephalopods, crustaceans, and oysters. Provisional Tolerable Weekly Intake (PTWI) suggested by Joint FAO/WHO Expert Committee on Food Additives (JECFA) for lead intake was 25 µg/kg b.w.
Conclusion
Analysis of toxic forms of methyl mercury and inorganic arsenic in seafood has to be done using precise and robust analytical methods with prime importance. Data of these toxic contaminants can give the human exposure level in different geographical locations and the risk associated.
(Laly, S.J., Jeyakumari, A. Abhay Kumar are scientists and Murthy L N is Principal scientist at the Mumbai Research Centre of ICAR-Central Institute of Fisheries Technology, Vashi, Navi Mumbai. Views expressed are personal. References can be provided on request.
